mRNA Vaccines and Therapeutics
Simplify your start to in vitro transcription
Developing next-generation therapeutics and vaccines using mRNA requires starting materials that meet the highest standards for yield and safety. Our opDNA® template offers a breakthrough solution removing complexity and risk to your manufacturing process compared to plasmid DNA.
Discover opDNA® template technology
The cell-free template that delivers increased yields, unparalleled purity and polyA stability
opDNA® is a construct with a 3’ stabilized open end, which feeds directly into in vitro transcription (IVT) reactions. This feature means it doesn’t require additional enzymatic linearization or de-ending steps, simplifying your mRNA manufacturing workflows. Thanks to our unique cell-free manufacturing process, long, continuous polyA tails can be directly encoded into the DNA template. This allows you to avoid the risk of batch failures and the need for additional enzymatic tailing steps, which is often seen with plasmid-based workflows.
Let our DNA experts customize a solution just for you.
opDNA® platform: engineered for performance and safety
Traditional mRNA production often relies on DNA templates derived from plasmid backbones, which introduce variability and regulatory risk, as well as enzymatic steps for linearization and polyA tailing.
Our unique opDNA® platform fundamentally improves the core of your mRNA synthesis:
Highest Purity, Unmatched Safety: Unlike competitors that rely on plasmid backbones, our unique cell-free manufacturing process eliminates the risk of host-cell contamination and removes antibiotic resistance genes. This ensures the highest safety and regulatory compliance for your advanced therapeutic products.
Streamline Manufacturing: The opDNA® construct comes with a 3’ open-end, allowing you to simply start your IVT reactions without the need for further optimization or enzymatic steps.
Increased Yields: This optimized, high-fidelity template has been shown to increase mRNA yields with less DNA input compared to traditional plasmid DNA, allow you to manage cost and timelines.
Integrated PolyA Sequence: Encode long, continuous polyA sequences directly into the template and remove the need for a separate, error-prone enzymatic tailing step, simplifying your workflow and boosting consistency.
Critical quality attributes that matter
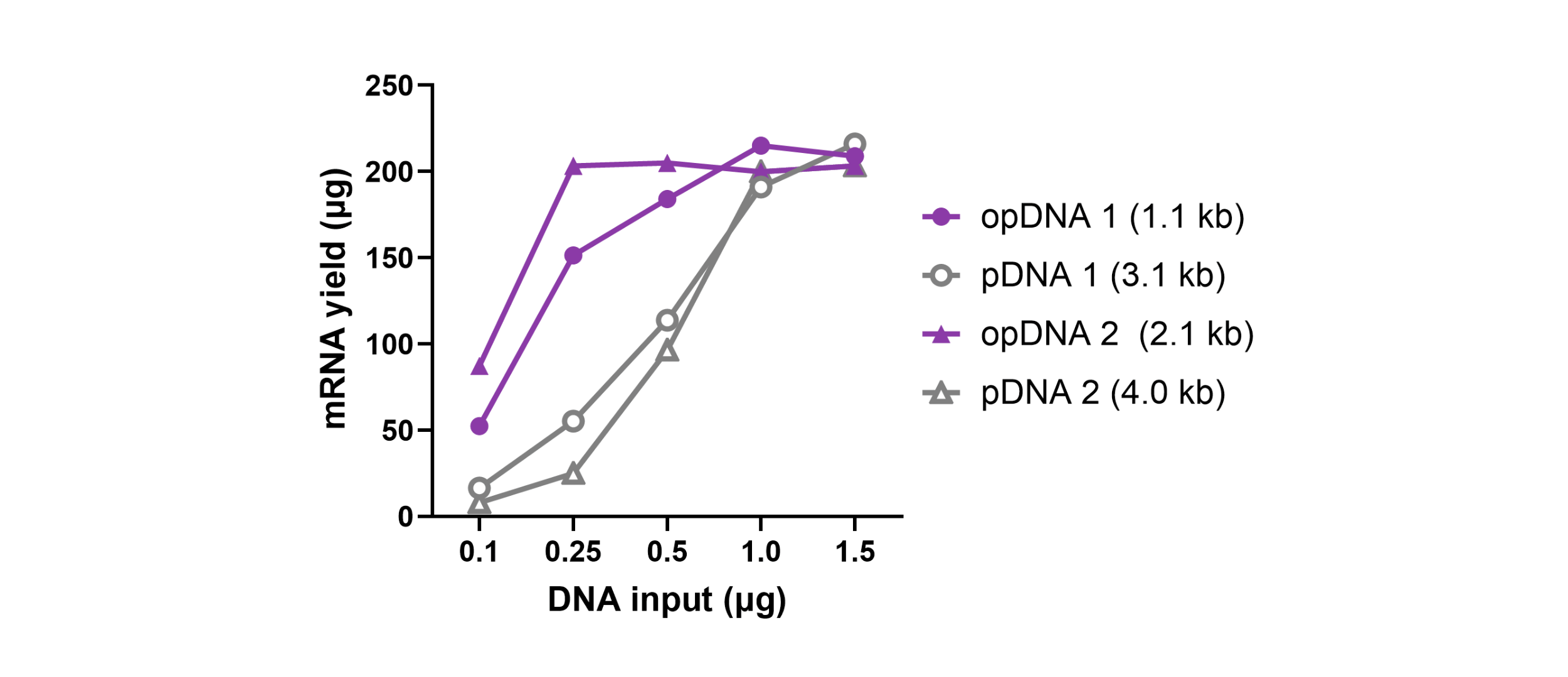
Achieve equivalent IVT mRNA yields at lower DNA inputs
Enhancing efficiency and reducing costs of your IVT reactions is crucial for mRNA therapeutic development. When compared to plasmid DNA, opDNA® achieves improved IVT yields for equivalent DNA mass. This is driven by the lack of bacterial backbone sequences in opDNA®, which only contain the sequence of interest from promoter to polyA tail.
Reduce dsRNA content vs traditional pDNA templates
Double-stranded RNA (dsRNA) is a major impurity in mRNA manufacturing, it can trigger strong innate immune responses, leading to reduced therapeutic efficacy and increased reactogenicity. dsRNA levels were measured using the J2 monoclonal antibody–based dot blot assay showing comparable levels of dsRNA between opDNA® and plasmid DNA-derived mRNA.
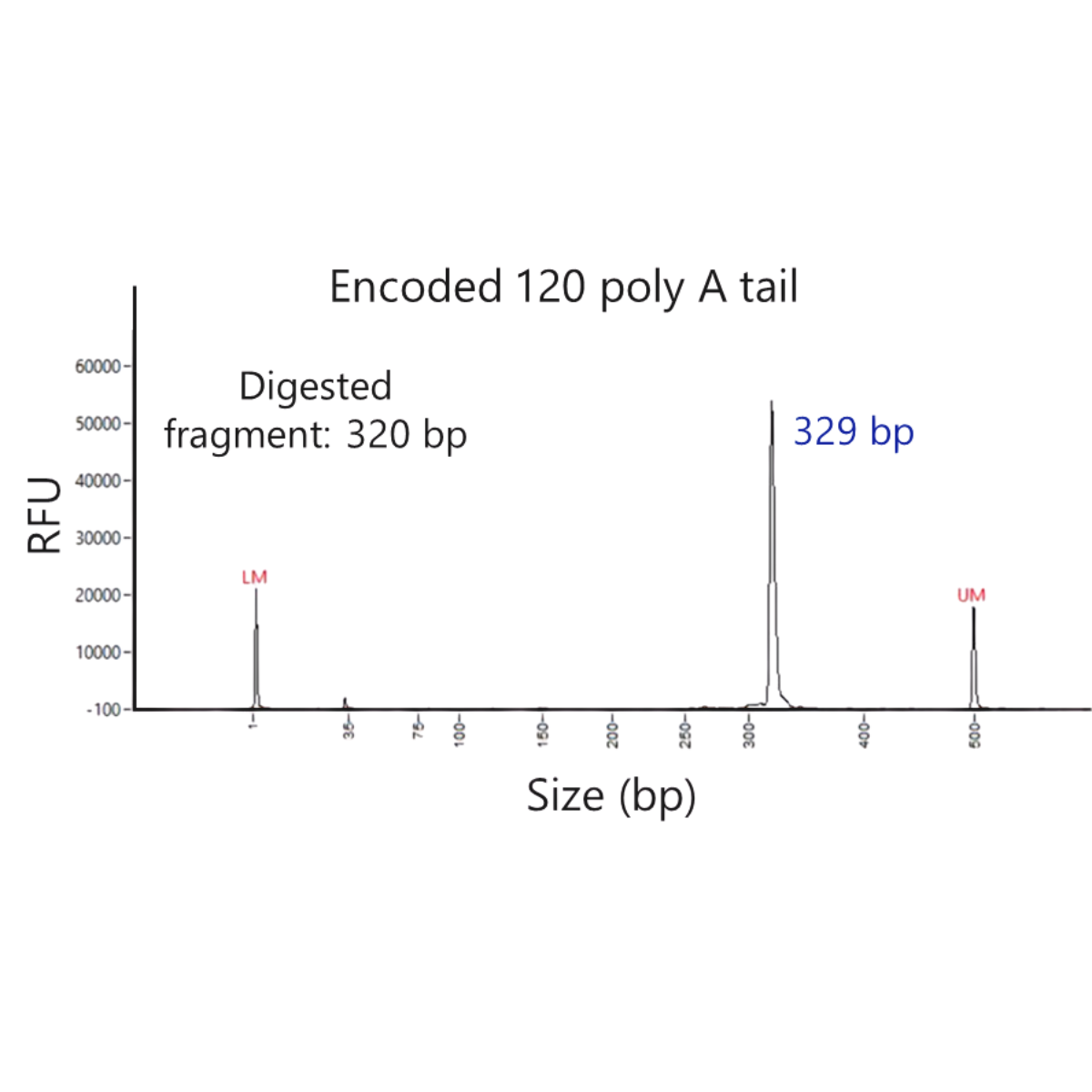
Improve PolyA stability with opDNA® templates
PolyA stability is often a challenge in fermentation-based manufacturing processes, 4basebio’s cell-free manufacturing allows for long polyA tails to be directly encoded into the DNA template. A full length polyA tail of up to 180bp can be encoded into the template and be fully maintained through amplification. A single peak of the expected length can be detected by capillary gel electrophoresis (+/- 5%).
Learn more about opDNA® technology with our recent whitepaper
opDNA FAQs
Your Questions, Answered
-
4basebio’s proprietary opDNA platform refers to protected ends linear DNA templates that have been specifically designed and synthesized for in vitro transcription (IVT) of mRNA with the 3’ end open for run off transcription. opDNA does not require any post-synthesis processing and is directly IVT compatible, whereas traditional plasmid DNA is circular requiring linearization and purification prior to run off transcription. For large scale IVTs with higher template requirements the restriction enzyme requirement for plasmid linearization may be a significant factor in the production cost. Plasmid DNA contains bacterial sequences contributing to both lenght of the IVT template and potential contamination risk downstream. With opDNA, only the GOI to be transcribed and RNA polymerase promoter are required allowing for a shorter template and therefore less template required per IVT on an equimolar basis. Additionally, opDNA synthesis can incorporate high quality long stretches of homopolymer sequences not at risk of mutations by recombination events like in plasmids, including up to 180 nt long polyadenine tail that can aid mRNA product stability and longevity.
-
Synthetic DNA offers superior consistency, purity, and scalability compared to PCR-amplified templates. It eliminates batch-to-batch variability associated with PCR-enzymatic amplification, reduces the risk of sequence errors that can accumulate through PCR cycles, and allows for highly scalable production without limitation on construct length. The 4basebio process uses proprietary in-house high fidelity enzymes for DNA amplification resulting in (200x?) greater sequence fidelity over PCR. Additionally, 4basebio platform has increased stability profile while simultaneously resistant against exonuclease digestion through modified ends. Altogether this provides a more reliable source material for GMP manufacturing of therapeutic mRNAs like vaccines or protein replacement therapies.
-
4basebio’s opDNA platform significantly influence mRNA immunogenicity by excluding unnecessary bacterial sequences, optimizing UTRs to reduce unintended immune activation, and enabling incorporation of modified nucleotides (like pseudouridine or N1-methylpseudouridine) during transcription that reduce recognition by innate immune sensors.
-
Plasmid DNA is circular and typically contains bacterial origin of replication, antibiotic resistance genes, and other sequences required for propagation in bacterial hosts. 4basebio’s proprietary enzymatic manufacturing process synthesizes a linear DNA template, which contains the transcription-essential elements: promoter, the gene of interest, and termination/poly(A) signals.
-
Circular plasmid DNA must be linearized with restriction enzymes downstream of the poly(A) sequence to create a defined transcription endpoint; otherwise, RNA polymerase continues around the plasmid, generating aberrant, long transcripts. This linearization step adds time, cost, and potential for incomplete digestion. 4basebio’s synthetic DNA is pre-designed with a 3' open end, eliminating the linearization step entirely, reducing process complexity and the risk of producing heterogeneous mRNA products.
Applications
Find out more about our enzymatic manufacturing process and the benefits of synthetic DNA.
Custom Manufacturing
Discover our RUO, HQ, and GMP-grade synthetic DNA, designed for every stage, from discovery to commercial production