
Personalized Cancer Vaccines and Therapies
Perfectly personalized. Cell-free solutions.
In the race to deliver personalized oncology, traditional DNA manufacturing is the bottleneck. 4basebio provides a high-purity, cell-free alternative to legacy processes, delivering the consistency and timelines your therapeutics demand.
High-purity, synthetic DNA for faster “batch-of-one” therapies
Faster. Safer. More Cost-Effective.
Traditional plasmid manufacturing is slow and carries the risk of bacterial contamination. Our proprietary enzymatic platform eliminates these hurdles, providing a streamlined path from design to delivery.
4basebio collapses these cycles into a reliable 6-week turnaround*, enabling our partners to meet the most aggressive clinical timelines and ensuring patient-specific treatments reach those who need them most.
Reliable 6-week turnaround*
Patient-specific readiness
High-purity synthetic DNA
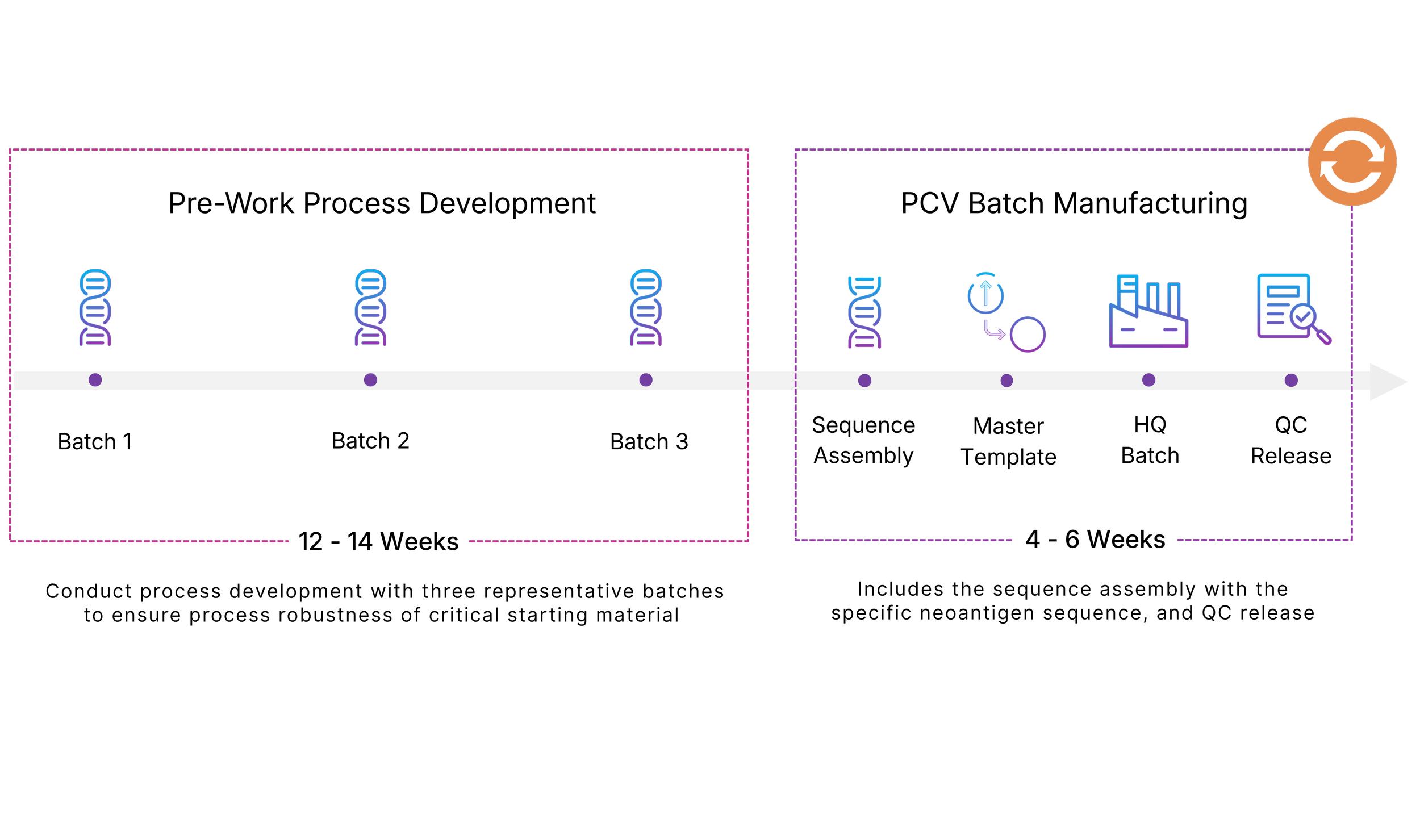
*This timeline can be achieved with prior process development steps over a 12 - 14 week period
When every patient is unique, your DNA supply shouldn’t be generic.
Let our DNA experts customize a solution just for you.
Optimized performance for next-gen oncology
High-fidelity, linear DNA templates feature unique hairpin structures and protected 3’ and 5’ ends, offering a cleaner, safer, and more efficient alternative to legacy plasmids.
We don’t just manufacture DNA; we optimize it for clinical success. Our synthetic DNA platform is engineered for the high-fidelity requirements of advanced biopharma.
The Cell-Free Advantage
High-Fidelity PolyA Tails: Our templates include encoded PolyA tails (up to 120bp+), ensuring superior mRNA integrity and potency.
Higher Yields, Lower Mass: The absence of bacterial backbones means you need less DNA per reaction, drastically reducing raw material costs.
Rapid Turnaround Times: With prior process development steps, we enable ~6-week timelines from biopsy to DNA supply, supporting the speed required for PCV workflows
No Bacterial Sequences Amplified: Our process starts from the production of a Master Template which is free-from bacterial sequences, which is then amplified in our enzymatic process
Small batch sizes: Custom small-batch DNA production optimized to you personalized therapy requirements
When patients are waiting, every day counts. 4basebio enables partners to meet aggressive clinical timelines with the speed and precision that modern oncology demands.
From tumor sequencing to treatment
Accelerating the journey from design to delivery with high-purity synthetic DNA
Each step of your PCV workflow is highly time-sensitive, and delays can compound across the workflow, directly affecting when patients receive treatment. To ensure you project is progressing smoothly, 4basebio has developed a specialized workflow to help achieve your required 6-week timeline.
Explore our cell-free DNA for PCV applications
Stop waiting on legacy bottlenecks
Start building the future of personalized medicine
Ready to accelerate your pipeline?
Technology
Find out more about our enzymatic manufacturing process and the benefits of synthetic DNA.
Custom Manufacturing
Discover our RUO, HQ, and GMP-grade synthetic DNA, designed for every stage, from discovery to commercial production
